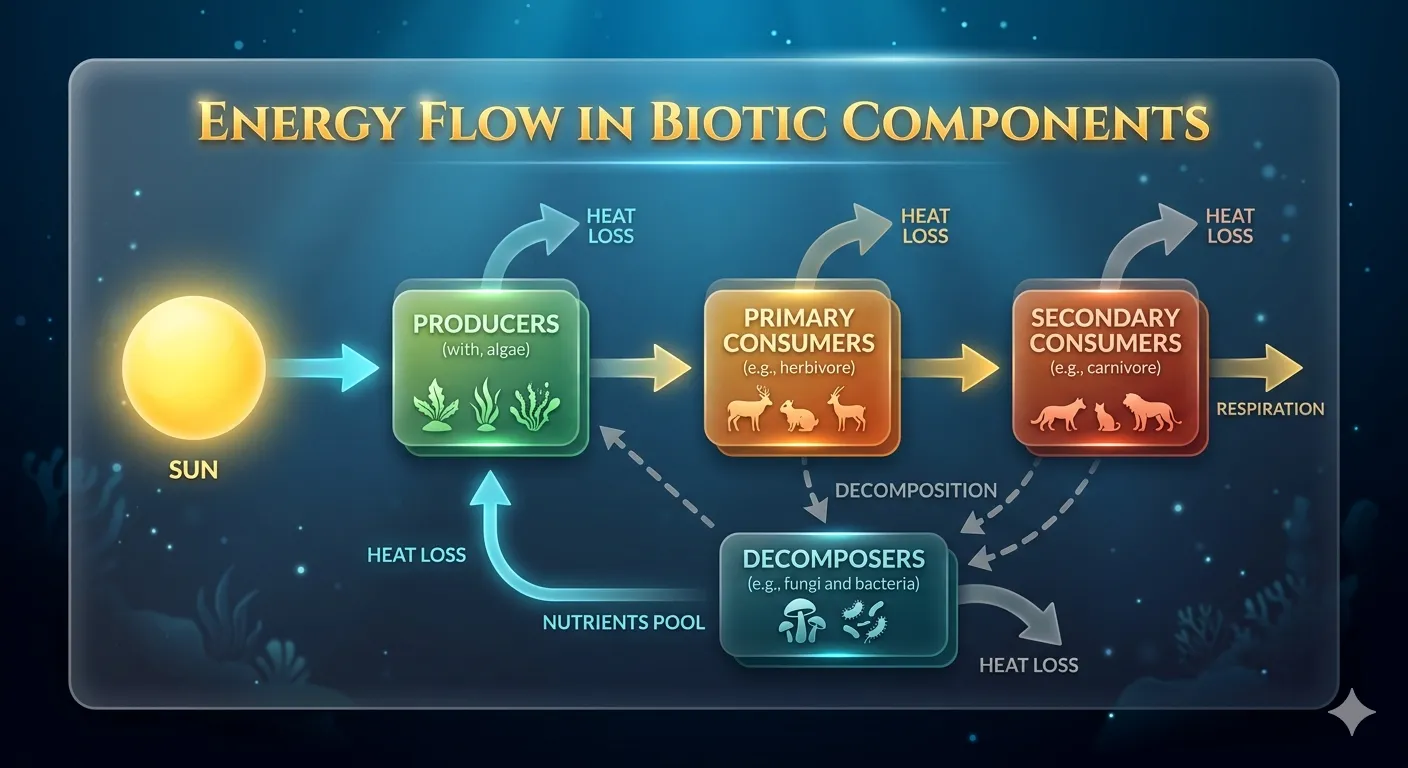
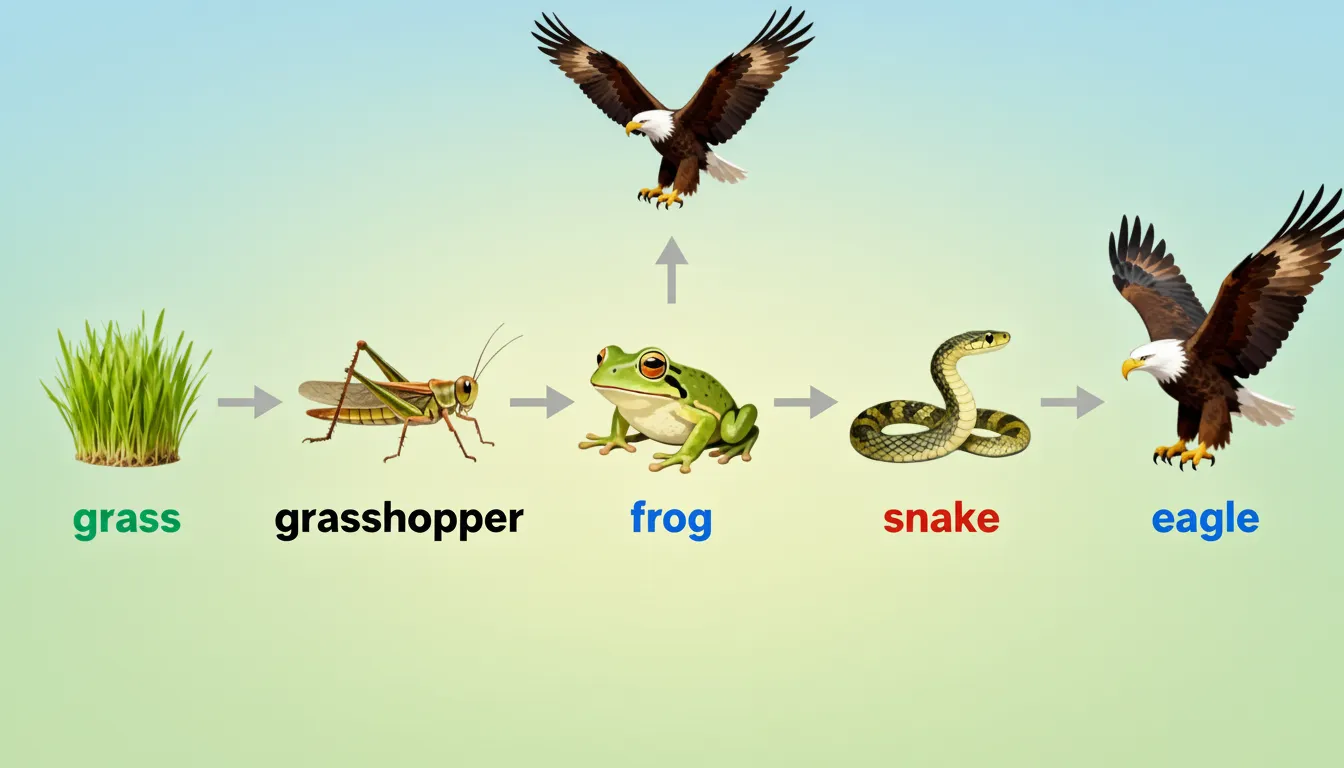
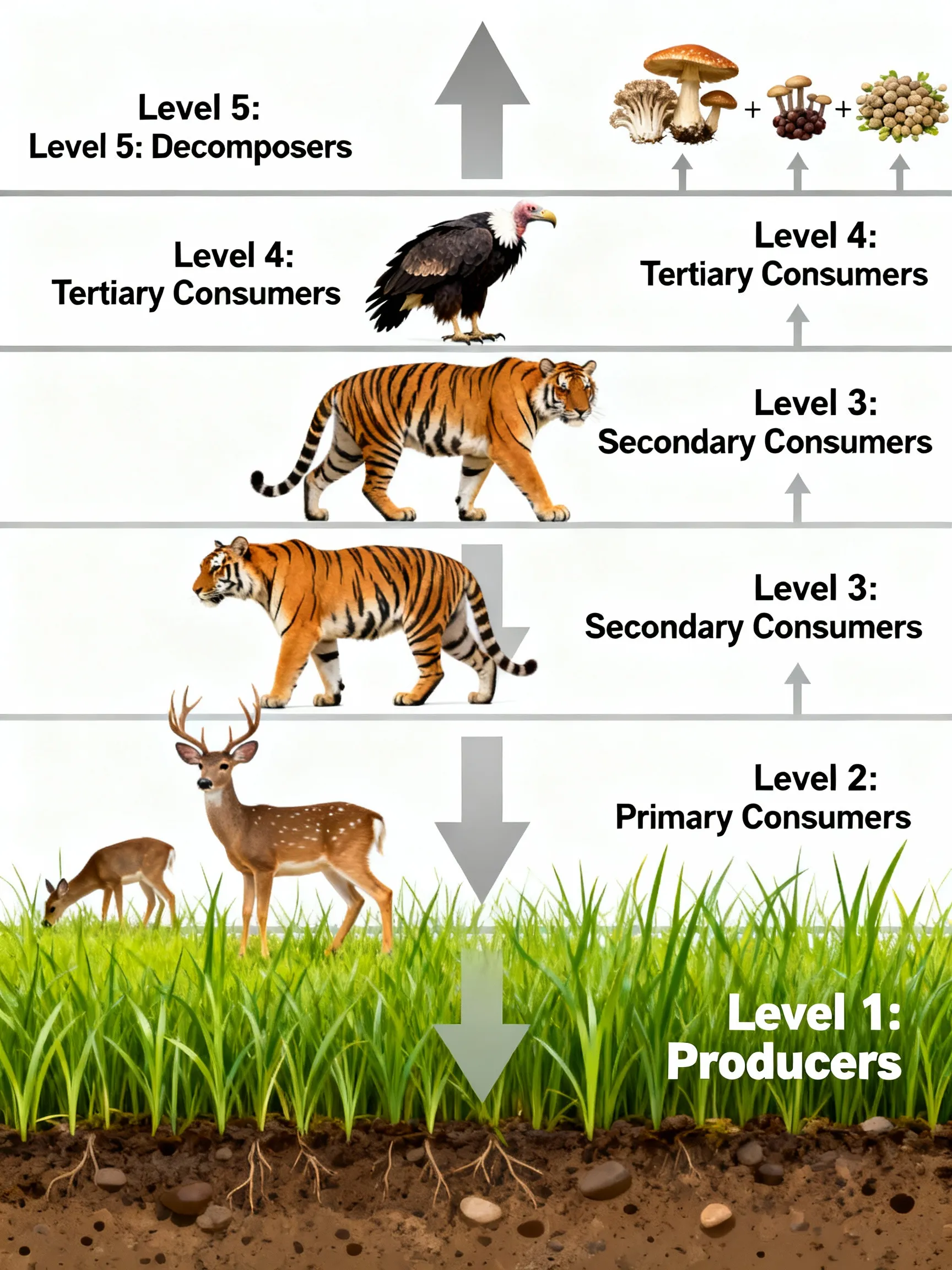
⚡ Energy Transfer — 10% Law (Lindemann)
Energy at Tₙ₊₁ = Energy at Tₙ × (10 / 100)
Energy at T₂ = Energy at T₁ × 0.1
Where T₁ = Producer level, T₂ = Herbivore level, etc. Only 10% of energy is transferred; 90% is lost as heat, respiration, locomotion.
🧮 Energy Calculation Examples
If T1 has 10,000 J → T2 = 1,000 J → T3 = 100 J → T4 = 10 J
If T3 has X joules → T1 = X × 100 joules (work backwards: multiply by 10 each step)
Energy Lost at each level = 90% of that level's energy
🌿 Photosynthesis (Producer Equation)
6CO₂ + 6H₂O → C₆H₁₂O₆ + 6O₂ (in presence of sunlight + chlorophyll)
🛡 Ozone Chemistry
O₂ + UV → O + O (photodissociation)
O + O₂ → O₃ (ozone formation)
O₃ + Cl• → ClO + O₂ (destruction by CFC-derived Cl)
ClO + O• → Cl + O₂ (Cl is regenerated — catalytic cycle!)
Net effect: O₃ is destroyed and Cl acts as a catalyst (not consumed), so one Cl atom can destroy up to 100,000 O₃ molecules.
📊 Biomagnification Relationship
Concentration at Tₙ ≈ Concentration at T₁ × 10^(n-1)
Chemical concentration multiplies by roughly 10× at each trophic level (inverse of energy transfer — energy goes down, chemical concentration goes up).
🔄 Key Ratios to Remember
| Quantity | Value / Relationship | Context |
|---|
| Energy transfer efficiency | 10% | Between each trophic level |
| Energy loss per level | 90% | Lost as heat + metabolism |
| Max food chain length | 4–5 levels | Due to 10% rule limiting energy |
| Ozone height in stratosphere | 15–35 km | Above Earth's surface |
| CFC destructive power | 1 Cl → 100,000 O₃ | Catalytic destruction |
| Ozone formula | O₃ | Triatomic oxygen molecule |