(a) 6 covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
Concept Required
- Carbon is tetravalent → it forms 4 covalent bonds.
- A covalent bond is formed by sharing of one pair of electrons.
- Single bond = 1 shared electron pair.
Solution Roadmap
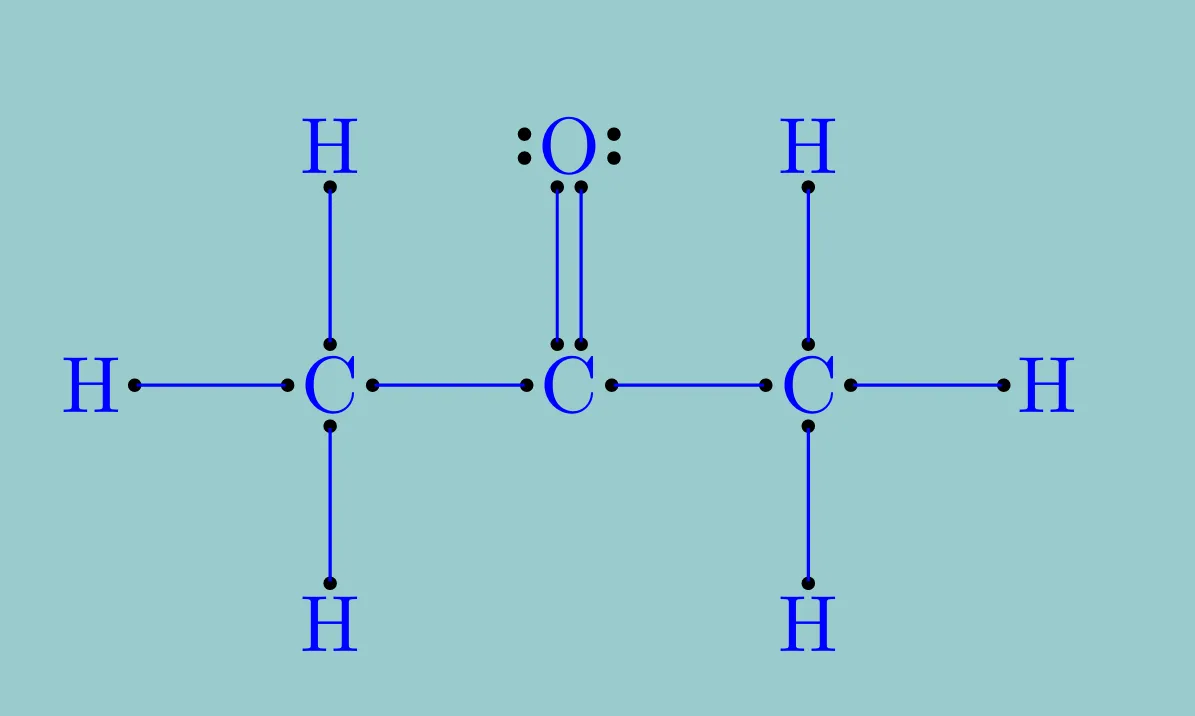
- Write the structure of ethane \(\ce{C2H6}\).
- Identify all bonds formed by each carbon atom.
- Count C–C and C–H bonds separately.
- Add total number of covalent bonds.
Step-by-Step Solution
Step 1: Write molecular composition
\[
\ce{C2H6}
\]
Ethane contains 2 carbon atoms and 6 hydrogen atoms.
Step 2: Use tetravalency of carbon
Each carbon atom forms 4 covalent bonds.
Step 3: Formation of C–C bond
The two carbon atoms share one pair of electrons:
\[
\ce{C - C}
\]
So, number of C–C bonds = 1
Step 4: Formation of C–H bonds
Each carbon is bonded to 3 hydrogen atoms:
\[
3 + 3 = 6 \text{ C–H bonds}
\]
Step 5: Total number of covalent bonds
\[
\text{Total bonds} = 1 + 6 = 7
\]
Final Answer: (b) 7 covalent bonds
Exam Significance
- Frequently asked in CBSE board MCQs and assertion-reason questions.
- Important for understanding structural formulas of hydrocarbons.
- Forms the base for higher topics like homologous series and isomerism.
- Helpful in competitive exams (NEET/JEE foundation level) for bond counting and valency concepts.